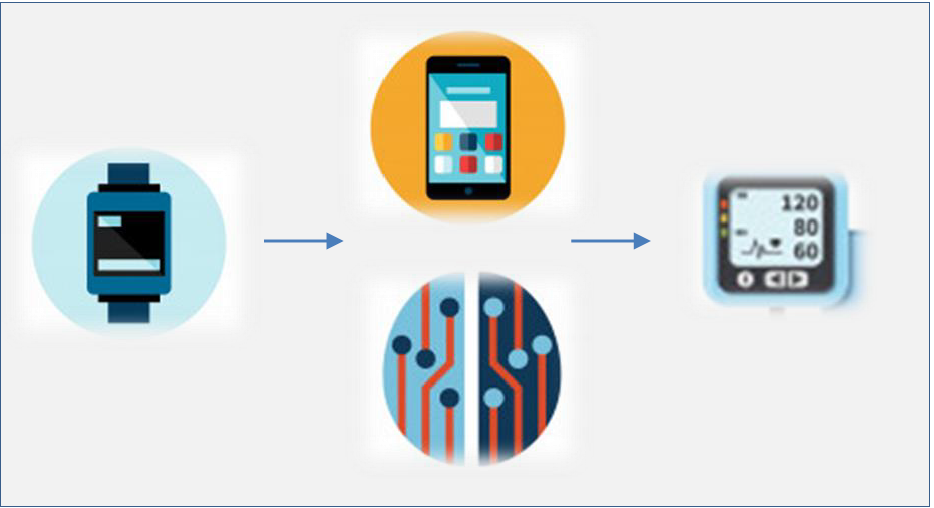
Ecg Watch looks like a common wristwatch, but able to detect an electrocardiogram, which can be viewed on smartphone and shared with the cardiology service center. There are no adhesive contacts or wires and the analysis can be performed even when standing up, without the need to undress. The App available on Smartphones displays the ECG, and is able to detect both the heart rate and atrial fibrillation.