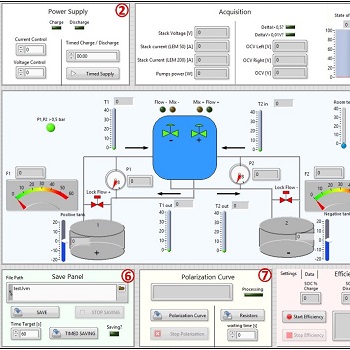
This new battery management system software is capable of interacting with the different hardware elements to which it is connected: signal conditioning system, power management system, hydraulic circuits, stack voltage and current, OCV etc.
By acquiring a multitude of parameters (open circuit voltage OCV, cell and stack voltage, input and output stack temperature, stack current, electrolyte flow rates, stack pressure drops, tank levels, pump power) the patented method behind the BMS can control battery functions, in particular:
- Identify a period of inactivity of the battery, during which it does not either accumulate or supply power;
- Activate an operational mode to maintain the accumulated charge during stand-by that:
- Interrupts the electrolyte flow from the stack to the battery or maintains a minimum flow necessary to guarantee adequate battery response time;
- identifies the threshold condition at which salts can precipitate within the stack; consequently activates a washing cycle that pumps fresh electrolyte solution from the tanks. This action is particularly important because in controls the stack temperature and avoids overheating.